Mechanosensing, Arteriogenesis and Ischemic Cardiovascular Diseases
| Arteriogenesis, the outward remodeling of pre-existing small collateral arterial networks, occurs as a response to vascular occlusion or stenosis and importantly determines the clinical outcome of ischemic cardiovascular disease. Release of vasodilators and activation of inflammatory pathways allowing influx of monocytes may result in revascularization and restoration of blood flow into the hypoperfused ischemic area. Therapeutic arteriogenesis is considered of major clinical importance to treat the increasing population with complex occlusive artery diseases. Distinct differences exist between animal strains and patients with regard to collateral development and response to angiogenic growth factors. We focus on determinants of native collateral number, and shear induced outward remodelling. |
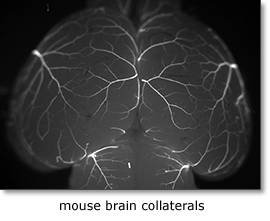 |
We aim at understanding the molecular mechanism accounting for the differences in native collateral network size. In particular we focus on the formation of native collaterals and efficiency of collateral recruitment and maintenance. We recently showed that the Notch-Dll4 signalling pathway acts a negative regulator of collateral network size (Cristofaro et al, Development 2013), and are currently exploring the therapeutic potential of these findings.
Hemodynamic forces exerted by flowing blood and neural guidance molecules (neuropilins-VEGFR2; Notch-delta ligands, ephrins and downstream targets) play a critical role in initiation and maintenance of the arteriogenesis response. We are interested in how biophysical signals exerted by flowing blood activate specific genetic programs essential for arterial differentiation, and recruitment of blood borne monocytes/macrophages allowing outward remodelling of collateral arteries (Hillmeister et al, Circ Res 2011).
The role of neural guidance molecules in the arteriogenesis is studied in conditional mutant mice exposed to ischemic vascular stress (femoral artery occludion, cardiac infarct, stroke models). We examine adaptive recovery from such pathological insults using laser-doppler flow, perfusion-microspheres, imaging including standard angiography, MRI and micro CT substantiate by molecular analysis of factors involved in arterial specification, and smooth muscle cell function.
Publications
- Hinkel R., Trenkwalder T., Petersen, B., Husada W., Gesenhues F., Lee S., Hannappel E., Bock-Marquette I., Theisen D., Leitner L., Boekstegers P., Cierniewski C., Müller O., Le Noble F., Adams R., Weinl C., Nordheim A., Reichart B., Weber C., Olson E., Posern G., Deindl E., Niemann H., and Kupatt C. (2014). MRTF-A controls vessels growth and maturation by increasing the expression of CCN1 and CCN2. Nature Communications. 5:3970. DOI:10.1038/ncomms4970
- Le Noble F., and Le Noble J. (2014). Bone biology: Vessels of rejuvenation. Nature 507, 313-4.
- Cristofaro B., Shi Y., Faria M., Suchting S., Leroyer A., Trindade A., Duarte A., Zovein A., Iruela-Arispe M., Nih L., Kubis N., Henrion D., Loufrani L., Todiras M., Schleifenbaum J., Gollasch M., Zhuang Z., Simons M., Eichmann A., and Le Noble F. (2013) Dll4-Notch signaling determines the formation of native arterial collateral networks and arterial function in mouse ischemia models. Development 140, 1720-9
- Jiang Q, Lagos-Quintana M, Liu D, Shi Y, Helker C, Herzog W, le Noble F. (2013) miR-30a regulates endothelial tip cell formation and arteriolar branching. Hypertension 62, 592-8
- Dimova I., Hlushchuk R., Makanya A., Styp-Rekowska B., Ceausu A., Flueckiger S., Lang S., Semela D., Le Noble F., Chatterjee S., and Djonov V. (2013). Inhibition of Notch signaling induces extensive intussusceptive neo-angiogenesis by recruitment of mononuclear cells. Angiogenesis 16, 921-37
- Napp, L.C., Augustynik, M., Paesler, F., Krishnasamy, K., Woiterski, J., Limbourg, A., Bauersachs, J., Drexler, H., Le Noble, F., Limbourg, F.P. (2012). Extrinsic Notch ligand Delta-like 1 regulates tip cell selection and vascular branching morphogenesis. Circulation Research 110, 530-5
- Krueger, J., Liu, D., Scholz, K., Zimmer, A., Shi, Y., Klein, C., Siekmann, A., Schulte-Merker, S., Cudmore, M., Ahmed, A., and Le Noble, F. (2011). Flt1 acts as a negative regulator of tip cell formation and branching morphogenesis in the zebrafish embryo. Development 138, 2111-2120
- Hillmeister, P.*, Gatzke, N.*, Dulsner, A., Bader, M., Schadock, I., Hoefer, I., Hamann, I., Infante-Duarte, C., Jung, G., Troidl, K., Urban, D., Stawowy, P., Frentsch, M., Li, M., Nagorka, S., Wang, H., Shi, Y., Le Noble, F*., and Buschmann, I*. (2011). Arteriogenesis Is Modulated By Bradykinin Receptor Signaling. Circulation Research 109, 524-533. *equal contribution
- Klein, C., Mikutta, J., Krueger, J., Scholz, K., Brinkmann, J., Liu, D., Veerkamp, J., Siegel, D., Abdelilah-Seyfried, S., and Le Noble, F. (2011) Neuron navigator 3a regulates liver organogenesis during zebrafish embryogenesis. Development 138, 1935-1945
- Liu, D., Krueger, J., and Le Noble, F. (2011) The role of blood flow and microRNAs in blood vessel development. International Journal of Developmental Biology 55, 419-429
- Buschmann, I., Pries, A., Styp-Rekowska, B., Hillmeister, P., Loufrani, L., Henrion, D., Shi, Y., Duelsner, A., Hoefer, I., Gatzke, N., Wang, H., Lehmann, K., Ulm, L., Ritter, Z., Hauff, P., Hlushchuk, R., Djonov, V., van Veen, T., and Le Noble, F. (2010). Pulsatile shear and Gja5 modulate arterial identity and remodeling events during flow-driven arteriogenesis. Development 137, 2187-2196
- Crispi, F., Bijnens, B., Figueras, F., Bartrons, J., Eixarch, E., Le Noble, F., Ahmed, A., and Gratacos, E. (2010) The shunt problem: control of functional shunting in normal and tumour vasculature. Circulation 121, 2427-2436
